Ergothioneine Supplier: How to Choose a Reliable Manufacturer in 2026?
2026-05-03 23:42:20
Why Choosing the Right Ergothioneine Supplier Matters More Than Ever in 2026
The global demand for ergothioneine powder supplier solutions has grown steadily over the past few years, driven by increasing interest in functional ingredients across nutrition and cosmetic industries. As more brands incorporate ergothioneine powder into formulations, the gap between high-quality and inconsistent supply sources has become more apparent. For buyers, this is no longer just a sourcing decision—it is a strategic one.
From a procurement perspective, selecting a qualified ergothioneine manufacturer directly affects product consistency, formulation performance, and long-term supply stability. In bulk purchasing scenarios, especially for bulk ergothioneine, variations in purity, production methods, and quality systems can significantly impact downstream applications. This is particularly relevant for companies operating in regulated or highly competitive markets.
At CHEN LANG BIO TECH, we have seen a clear shift: buyers are moving beyond price-driven decisions and placing greater emphasis on supplier transparency, technical capability, and production reliability. Understanding how to evaluate a professional ergothioneine powder supplier is therefore essential for minimizing risk and ensuring consistent supply.
What Is Ergothioneine Powder and Why It Is in Demand
What is ergothioneine powder? It is a naturally occurring amino acid derivative widely used as a functional ingredient in both dietary supplement and cosmetic formulations. As a stable and highly water-soluble compound, ergothioneine ingredient solutions are valued for their compatibility across a wide range of product systems.
In industrial supply chains, ergothioneine raw material is typically produced through controlled fermentation or synthetic processes, depending on the manufacturer’s technology platform. This makes it suitable for standardized production, ensuring consistent batch quality for commercial use.
The increasing adoption of ergothioneine in supplements reflects a broader shift toward incorporating advanced antioxidant-related compounds into modern nutrition products. At the same time, the use of ergothioneine skincare ingredient formulations has expanded, particularly in applications where stability and formulation compatibility are critical.
From a sourcing standpoint, this growing demand has led to a more competitive supplier landscape. As a result, identifying a reliable ergothioneine powder supplier with strong technical and manufacturing capabilities has become a key priority for brands and contract manufacturers alike.
Key Factors When Choosing an Ergothioneine Supplier
Selecting the right supplier involves more than reviewing basic product specifications. For professional buyers, especially those sourcing bulk ergothioneine powder, a structured evaluation across quality, production, and supply factors is essential.
Product Purity and Specification Standards
One of the first aspects to assess is ergothioneine purity 98% vs 99%, as even small differences in purity levels can influence formulation consistency and cost efficiency. High-quality suppliers typically provide detailed ergothioneine specification documents outlining assay methods, impurity profiles, and testing standards. We mainly supply more than 99% ergothioneine powder, and supply ergothioneine COA and Third Party Test Data to control the quality and purity.
A verified ergothioneine coa (Certificate of Analysis) is a critical requirement in this process. It not only confirms batch-specific quality but also reflects the supplier’s testing capability and transparency. For buyers seeking consistent performance, working with a high purity ergothioneine supplier ensures alignment between specification claims and actual product quality.
At CHEN LANG BIO TECH, we recommend that buyers request full documentation and validate testing methods before making procurement decisions, especially when sourcing for large-scale production.
Manufacturing Process: Fermentation vs Synthetic
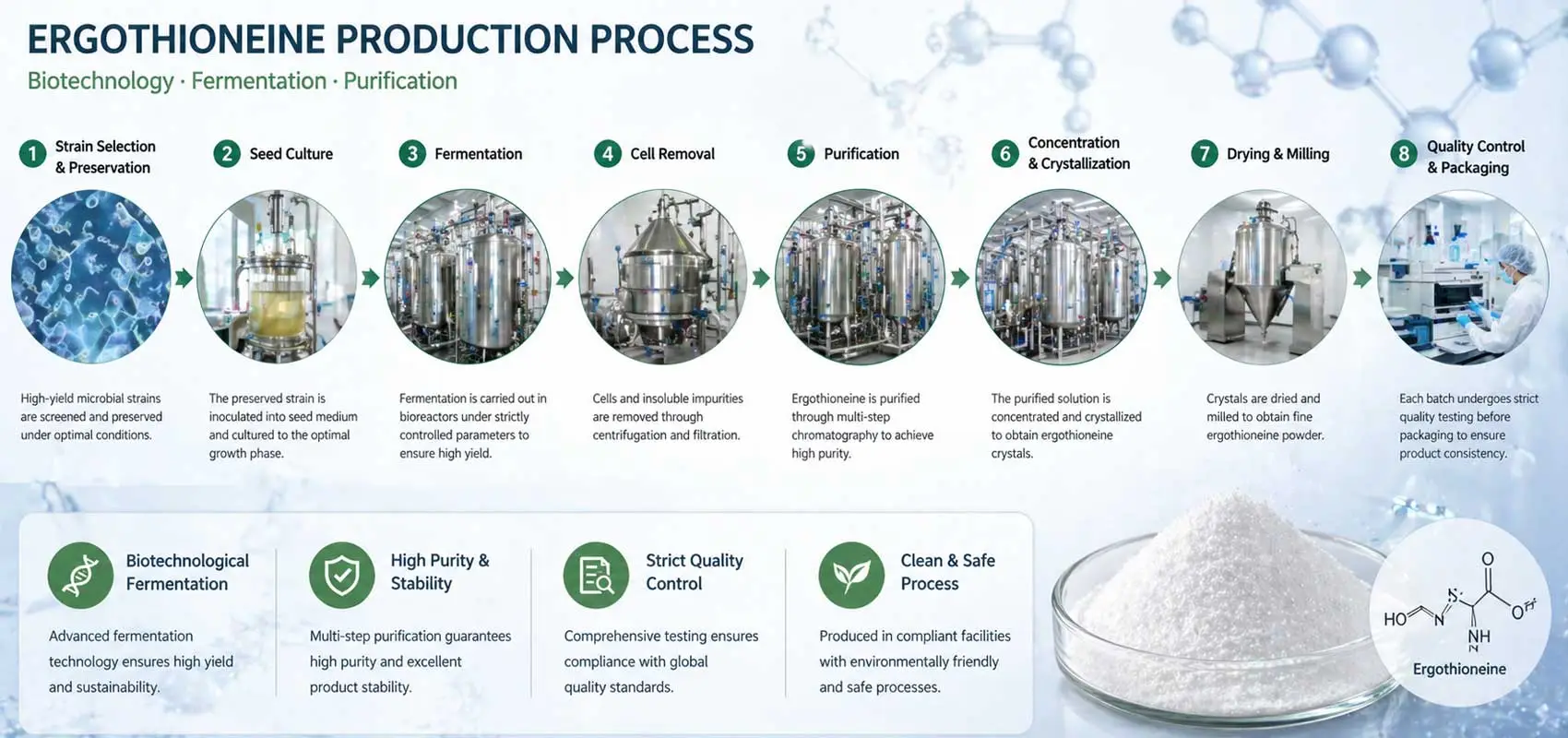
The production route plays a significant role in determining product quality and scalability. Today, most commercial ergothioneine fermentation processes are based on microbial synthesis, offering advantages in consistency and scalability. Alternatively, synthetic ergothioneine may be used in certain applications, depending on cost structure and production design.
Understanding the ergothioneine production process allows buyers to evaluate factors such as batch reproducibility, impurity control, and long-term supply stability. A qualified ergothioneine manufacturer should be able to clearly explain their production technology, including raw material sourcing and process controls.
From an industrial perspective, fermentation-based production is increasingly favored due to its alignment with scalable manufacturing and controlled quality output.
Supplier Certifications and Quality Control
Reliable suppliers typically operate under established quality systems. When evaluating an ergothioneine supplier certification, buyers should look for recognized standards such as ISO or GMP compliance, depending on the intended application.
A gmp ergothioneine manufacturer demonstrates structured production management, traceability, and standardized operating procedures. Beyond certifications, effective quality control ergothioneine processes—such as batch testing, stability monitoring, and documentation practices—are equally important.
For long-term partnerships, consistent quality performance is often more valuable than short-term pricing advantages. This is particularly true for brands requiring stable supply across multiple production cycles.
Supply Capacity and Bulk Availability
For commercial-scale operations, supply capability is a critical factor. A dependable supplier should be able to provide bulk ergothioneine powder with consistent availability, supported by clear production planning and inventory management.
Key considerations include ergothioneine bulk supplier capacity, minimum order quantities (ergothioneine MOQ), and delivery timelines. Understanding ergothioneine lead time is especially important for companies managing complex supply chains or seasonal demand cycles.
At CHEN LANG BIO TECH, we emphasize transparent communication on production scheduling and logistics, ensuring that clients can plan procurement with confidence.
Pricing Transparency and Cost Factors
While pricing remains an important consideration, it should be evaluated in context. The ergothioneine price per kg can vary depending on purity level, production method, and order volume. Buyers comparing ergothioneine bulk price options should also consider factors such as consistency, documentation, and supplier reliability.
Understanding the overall l-ergothioneine cost structure helps avoid common sourcing risks, such as inconsistent quality or hidden operational costs. A professional supplier should provide clear and transparent ergothioneine supplier pricing, allowing buyers to make informed decisions based on total value rather than unit price alone.
Ergothioneine Powder Applications: What Buyers Should Know
From a formulation perspective, ergothioneine supplement ingredient and cosmetic applications continue to expand, driven by its stability and compatibility with other functional components.
Use in Dietary Supplements
In the nutrition sector, bulk ergothioneine for supplements is typically used in capsule, tablet, or powder formulations. Manufacturers often evaluate ergothioneine dosage ranges based on formulation goals and regulatory considerations.
For brands, sourcing a consistent ergothioneine supplement ingredient is essential to ensure product uniformity and quality across production batches.
Use in Skincare and Cosmetics
In cosmetic formulations, ergothioneine in skincare is valued for its stability and compatibility with other active ingredients. As a cosmetic grade ergothioneine, it can be incorporated into serums, creams, and emulsions without significant formulation challenges.
From a technical standpoint, understanding ergothioneine formulation behavior—such as solubility and stability—is important when designing finished products. This further highlights the importance of working with an experienced supplier capable of providing technical support alongside raw materials.
Red Flags: How to Identify an Unreliable Supplier
As demand for ergothioneine powder supplier options continues to grow, the market has also become more fragmented. Not all suppliers operate with the same level of manufacturing control or quality transparency, which makes early-stage evaluation increasingly important.
One of the most common warning signs is pricing that falls significantly below the typical ergothioneine price per kg. While competitive pricing is expected in bulk procurement, unusually low offers often reflect compromises in raw materials, process control, or final product purity. Inconsistent alignment between declared and actual ergothioneine specification is a risk that buyers should not overlook.
Documentation is another critical factor. A credible supplier should be able to provide a complete and verifiable ergothioneine coa, supported by clear analytical methods. When documentation is vague, outdated, or difficult to verify, it may indicate underlying ergothioneine quality issues that could affect downstream applications.
Batch consistency is equally important, especially for companies sourcing bulk ergothioneine powder. If there is a noticeable gap between initial samples and production batches, it often points to limitations in process standardization. This is frequently associated with low quality ergothioneine supply chains where quality control systems are not fully established.
In addition, communication style can reveal a great deal about a supplier’s capabilities. A reliable partner is typically transparent about their ergothioneine production process, responsive to technical questions, and realistic about timelines. On the other hand, unclear answers or overly aggressive sales positioning may suggest a fake ergothioneine supplier or a distributor without direct manufacturing oversight.
From our experience at CHEN LANG BIO TECH, taking the time to identify these risks early leads to more stable and predictable sourcing outcomes.
Ergothioneine Supplier Checklist
For buyers evaluating multiple ergothioneine supplier options, a structured approach can simplify comparisons and reduce uncertainty. Rather than focusing on a single factor, it is more effective to assess suppliers across several key dimensions.
Product quality should always come first. Differences in ergothioneine purity 98% vs 99% can influence both formulation performance and cost efficiency, particularly in large-scale production. A clear and detailed ergothioneine specification provides insight into assay methods, impurity control, and overall product consistency. Supporting documentation, including a recent ergothioneine coa, helps verify that the material meets the expected standard.
Equally important is understanding how the product is made. Whether based on ergothioneine fermentation or synthetic routes, the underlying ergothioneine production process determines scalability, batch consistency, and long-term supply stability. Working directly with an experienced ergothioneine manufacturer offers greater visibility into these factors than sourcing through intermediaries.
Certifications and internal quality systems also play a significant role. A supplier operating as a gmp ergothioneine manufacturer typically follows structured production and documentation practices, supported by defined quality control ergothioneine procedures. This level of organization becomes particularly valuable when scaling production or entering regulated markets.
Supply capability is another practical consideration. A dependable ergothioneine bulk supplier should be able to meet demand consistently, with clearly defined ergothioneine MOQ requirements and predictable ergothioneine lead time. For companies managing complex supply chains, reliability in delivery is just as important as product quality.
Finally, pricing should be evaluated in context. Variations in ergothioneine bulk price often reflect differences in purity, production methods, and operational scale. Understanding the broader l-ergothioneine cost structure allows buyers to make informed decisions based on long-term value rather than short-term savings. Transparent ergothioneine supplier pricing is usually a strong indicator of a supplier’s professionalism.
Why Work with a Professional Ergothioneine Manufacturer
As the market matures, the role of a qualified ergothioneine manufacturer goes beyond supplying raw materials. For many brands, especially those developing new formulations, a reliable ergothioneine powder supplier becomes a strategic partner rather than just a vendor.
Consistency is one of the most immediate advantages. High-quality ergothioneine powder must perform reliably across batches, particularly in applications where formulation stability is critical. Suppliers with established production systems are better positioned to maintain this level of consistency over time.
Technical support is another factor that is often underestimated. Experienced manufacturers can provide practical guidance on ergothioneine formulation, including considerations around solubility, compatibility, and storage. For companies working with bulk ergothioneine, this support can streamline both development and production processes.
Supply stability is equally important. A reliable ergothioneine supplier with scalable production capacity can offer consistent lead times and flexible ordering options, helping buyers manage inventory and reduce operational risk. This is particularly valuable for businesses operating across multiple markets or product categories.
At CHEN LANG BIO TECH, our approach is built on combining manufacturing expertise with transparent communication. By focusing on both product quality and long-term partnership, we aim to support clients in building stable and efficient supply chains.
Conclusion: Making a Smarter Choice in 2026
Choosing the right ergothioneine powder supplier requires a balanced understanding of both technical and commercial factors. From evaluating the ergothioneine production process to comparing the ergothioneine price per kg, each step contributes to a more informed sourcing decision.
For companies sourcing bulk ergothioneine manufacturer solutions, the focus should be on consistency, transparency, and long-term reliability. Suppliers that can clearly demonstrate their capabilities—through documentation, communication, and performance—are more likely to deliver stable results over time.
As demand continues to grow, building relationships with experienced partners becomes increasingly important. With the right approach, buyers can not only secure high-quality ergothioneine powder, but also strengthen their overall supply strategy in a competitive and evolving market.
In practice, many procurement teams find it useful to begin with small-scale validation—reviewing technical documents, comparing specifications, and requesting samples before moving into larger commitments. If you are currently evaluating options or refining your sourcing strategy, having an open technical discussion with a qualified ergothioneine manufacturer can often provide clarity beyond what is available on paper.
At CHEN LANG BIO TECH, we are always available to support these early-stage evaluations, whether through sharing detailed product information, discussing application considerations, or simply helping you compare different supply approaches based on your specific requirements.
Send Inquiry
Related Industry Knowledge
- How to Use Phycocyanin Powder in Gummies, Desserts, and Functional Foods?
- Is Bifida Ferment Lysate Good for Skin?
- Is Piroctone Olamine Better Than Zinc Pyrithione?
- The Role of 5HTP Powder in Expanding Health Product Portfolios
- Inside Scoop: Fucoidan Powder’s Role in UV Skin Defense
- Is Piroctone Olamine Powder the Future of Anti-Fungal Formulations?
- Copper PCA for Oily Skin: Is It the Perfect Solution?
- Meglumine Powder and Its Role in Modern Medicine
- Are There Any Solubility Considerations for Kopyrrol Powder?
- Pro-xylane Powder: Anti-Aging Miracle