Ashwagandha Extract Safety and Side Effects: Considerations for Supplement Applications
2026-03-26 16:00:12
What Is Ashwagandha Extract Powder and Why Safety Matters in Modern Formulations
Ashwagandha extract powder, derived primarily from the root of Withania somnifera, has become one of the most widely discussed botanical ingredients in contemporary supplement development. In its standardized form, it is used across stress, sleep, cognitive wellness, sports nutrition, and daily adaptogen blends because it offers more consistent active-compound delivery than raw botanical powder. That distinction is important: when formulators refer to what is ashwagandha extract powder, they are usually referring to a concentrated ingredient with defined specifications rather than a simple ground herb.
From a product-development perspective, safety is not a secondary issue. It sits alongside efficacy, standardization, and supply consistency as a core criterion in ingredient selection. This is especially true for withania somnifera extract powder used in modern formulations, where the finished product may be positioned for repeated daily use. Public health agencies note that ashwagandha appears to be generally well tolerated in the short term, but they also emphasize that long-term safety is still not fully established and that side effects, interactions, and rare serious events have been reported.
In practical terms, the discussion around herbal extract safety is no longer just about whether an ingredient is traditionally used. It is about whether the material is standardized, well-characterized, cleanly processed, and supported by documentation that makes its use appropriate for real-world supplement applications. For an ingredient such as ashwagandha extract powder, safety depends not only on dose, but also on extract type, withanolide content, manufacturing controls, and the quality profile of the final batch. That is why safety assessment should begin with the raw material itself, not only with the label claim on the finished product.
Common Side Effects of Ashwagandha Extract: What Does the Evidence Show?
Mild and Short-Term Reactions
Based on NIH and NCCIH summaries of clinical and safety data, the most commonly reported ashwagandha extract side effects are relatively mild and typically include stomach upset, loose stools or diarrhea, nausea, vomiting, and drowsiness. In clinical settings, ashwagandha has generally been described as well tolerated for short-term use, particularly over periods of about 6 to 12 weeks and up to roughly 3 months, depending on the preparation studied.
For formulators, this matters because mild gastrointestinal discomfort is one of the most relevant considerations when evaluating ashwagandha digestive issues in a finished supplement. These reactions do not necessarily indicate that the ingredient is unsuitable, but they do reinforce the need for thoughtful dose selection, appropriate positioning, and a well-defined extract specification. In other words, a responsible safety profile is usually built at the formulation stage rather than corrected afterward.
In the broader context of herbal supplement side effects, ashwagandha is not unusual in presenting mostly mild, transient reactions in the short term. What distinguishes it, however, is that it is often marketed across very different categories—from stress relief to sports recovery—so the same ingredient may be used by consumers with very different health backgrounds, medication use, and tolerance levels. That makes clarity around its side-effect profile especially important in product communication and specification review.
Rare but Reported Adverse Effects
A more serious part of the safety discussion concerns rare but documented liver injury cases linked to products containing ashwagandha. NCCIH and the NIH Office of Dietary Supplements both note that although such events appear to be uncommon, case reports have described cholestatic or mixed-pattern liver injury in some individuals using ashwagandha-containing supplements. In the cases summarized by ODS, reported intake ranged from 450 to 1,350 mg per day over periods from about 1 week to 4 months, though not all products were independently verified and some were combination products.
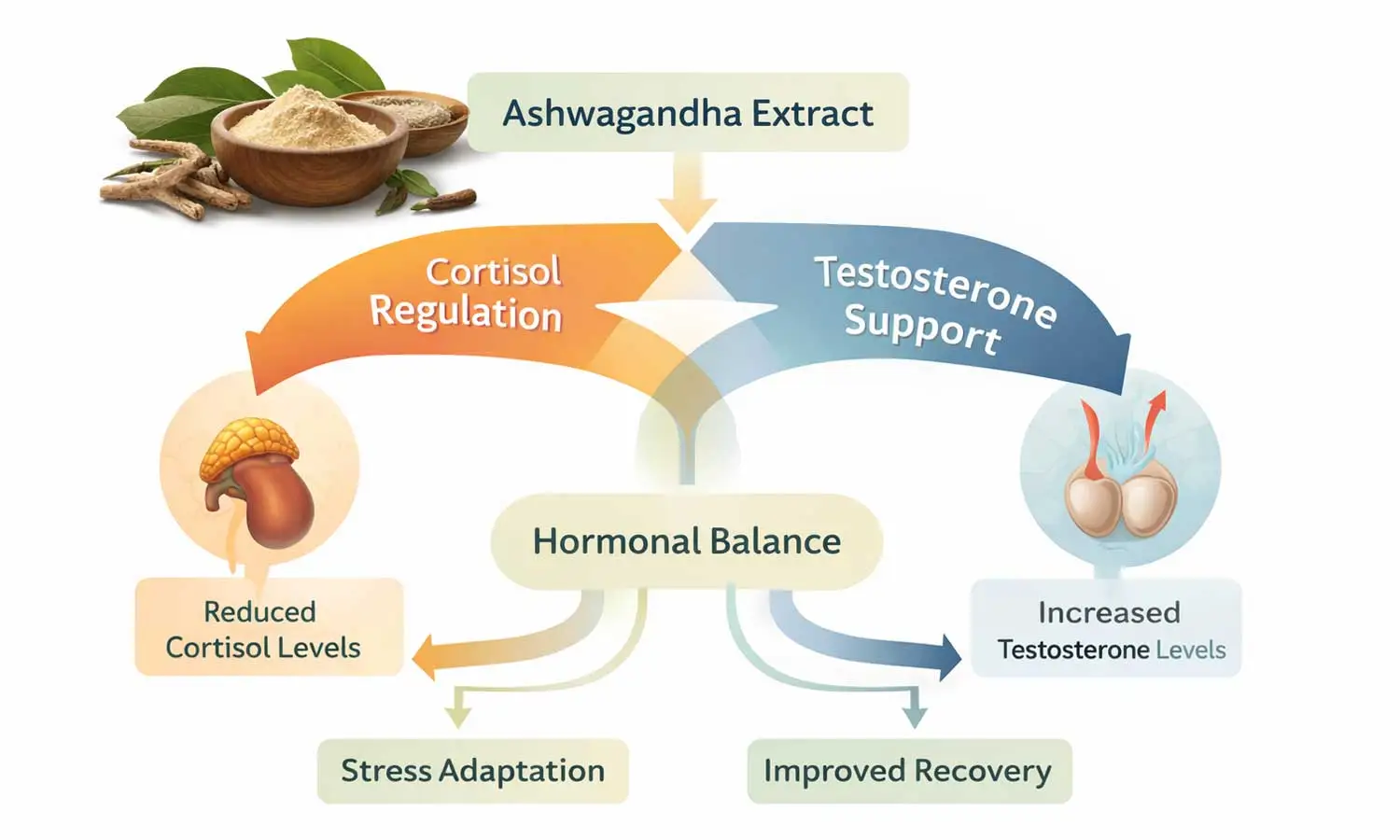
This is where the discussion around ashwagandha risks becomes more nuanced. The existence of rare adverse events does not automatically make the ingredient broadly unsafe, but it does mean that quality, traceability, and product composition matter. The available evidence also indicates that ashwagandha may affect thyroid function, may interact with sedatives, immunosuppressants, antihypertensives, antidiabetic medications, anticonvulsants, and thyroid medications, and should be avoided during pregnancy and breastfeeding. NCCIH also advises caution for people with autoimmune or thyroid disorders and for those preparing for surgery.
From an evidence-based standpoint, the answer to is ashwagandha safe long term is still incomplete. Federal health sources consistently state that short-term use appears reasonably well tolerated, but long-term safety remains insufficiently characterized. That is why the most credible adaptogen safety profile is one that is presented with appropriate boundaries: short-term human data are encouraging, but duration, product quality, and user-specific risk factors still matter.
Dosage, Standardization, and Safety: Why Withanolides Content Matters
Recommended Dosage Ranges in Supplement Applications
One of the most important principles in evaluating ashwagandha dosage extract safety is that dose cannot be separated from extract type. NIH’s Health Professional Fact Sheet summarizes clinical studies in which ashwagandha dosing ranged from 240 to 1,250 mg/day of extract, while one study used 12,000 mg/day of whole-root granules, equivalent to 6,000 mg of root powder. For sleep-focused trials, root extract doses commonly ranged from 250 to 600 mg/day, and for stress-related outcomes, benefits in several studies appeared more prominent around 500 to 600 mg/day.
In practical supplement design, that means there is no single universal “best” dose for safe dosage ashwagandha extract. A reasonable and conservative approach is to anchor dosing to the exact extract specification, withanolide standardization, target population, and intended duration of use. It is also worth noting that an international taskforce cited by ODS provisionally recommended 300 to 600 mg/day of root extract standardized to 5% withanolides for generalized anxiety disorder, while emphasizing that stronger evidence is still needed.
For supplement formulation dosage, what matters most is not simply the total milligrams on paper, but whether the dosage is technically matched to the extract profile and supported by tolerability data. Higher milligram amounts do not automatically mean better formulation, especially when withanolide content, excipient system, and user population are not clearly defined.
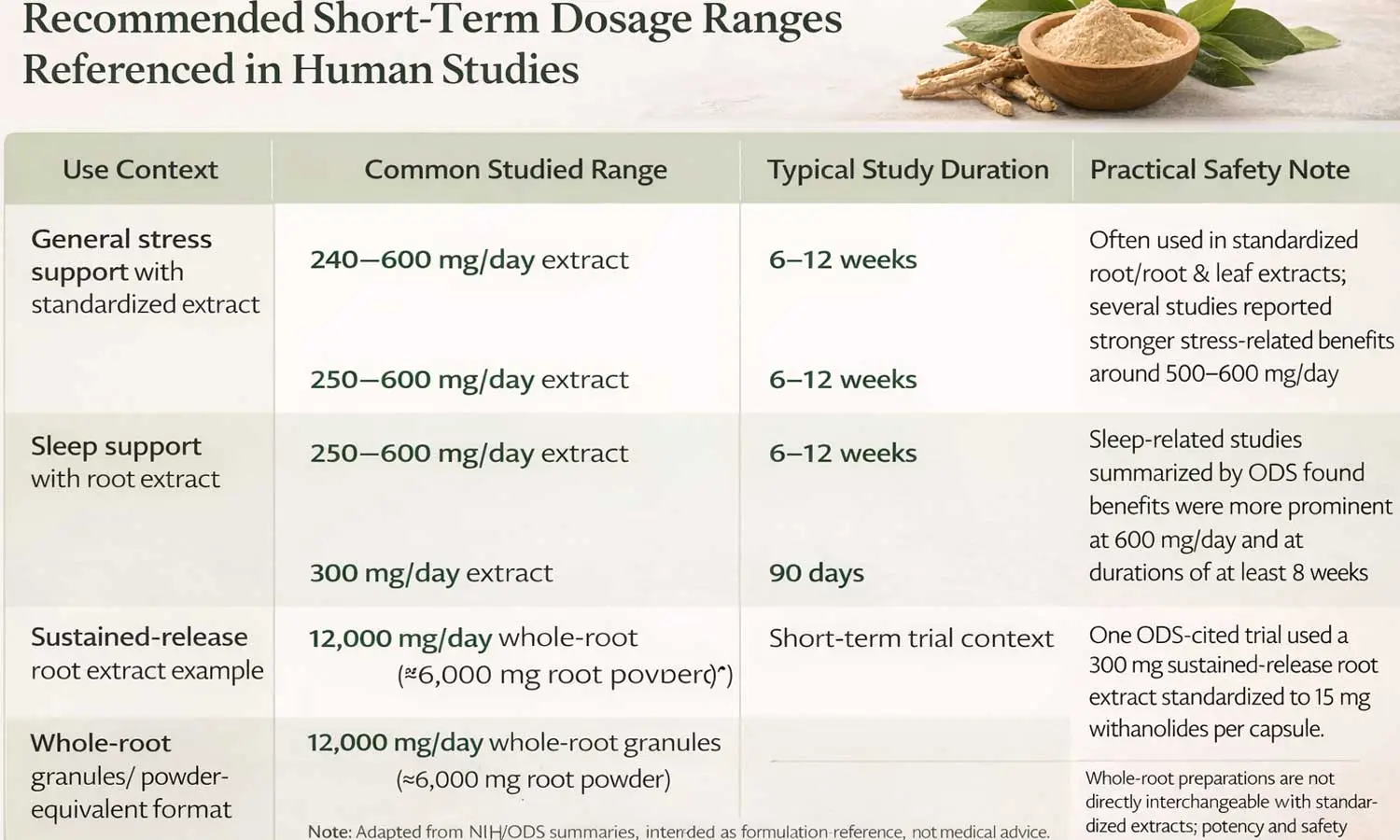
Recommended Short-Term Dosage Ranges Referenced in Human Studies
The table below summarizes dosage patterns commonly seen in short-term human studies and NIH summaries. It is designed as a formulation-oriented reference rather than a medical prescribing guide.
|
Use context |
Common studied range |
Typical study duration |
Practical safety note |
|
General stress support with standardized extract |
240–600 mg/day extract |
6–12 weeks |
Often used in standardized root or root/leaf extracts; several studies reported stronger stress-related benefits around 500–600 mg/day. |
|
Sleep support with root extract |
250–600 mg/day extract |
6–12 weeks |
Sleep-related studies summarized by ODS found benefits were more prominent at 600 mg/day and at durations of at least 8 weeks. |
|
Sustained-release root extract example |
300 mg/day extract |
90 days |
One ODS-cited trial used a 300 mg sustained-release root extract standardized to 15 mg withanolides per capsule. |
|
Whole-root granules / powder-equivalent format |
12,000 mg/day whole-root granules (≈ 6,000 mg root powder) |
Short-term trial context |
Whole-root preparations are not directly interchangeable with standardized extracts; potency and safety interpretation should not be assumed to match extract dosing. |
|
Thyroid-related clinical context |
300 mg twice daily |
8 weeks |
This dose appeared in a trial involving people with subclinical hypothyroidism, where thyroid markers changed; this should be treated as a cautionary data point, not a general wellness recommendation. |
Dosage and safety are closely related to extract concentration. For example, different withanolides levels can significantly influence formulation design, as discussed in our ashwagandha extract 5% vs 10% withanolides guide.
Standardized Extract vs Raw Powder: Safety Implications
The distinction between ashwagandha extract vs powder is highly relevant in safety assessment. A standardized extract is typically designed to deliver a reproducible amount of active compounds, especially withanolides, while raw powder reflects the whole-root material and may vary substantially in potency and composition. Because of that, standardized ashwagandha extract often gives formulators a more dependable basis for dose setting, quality control, and label consistency.
At the same time, the commonly discussed ashwagandha extract powder benefits—better potency control, lower capsule burden, and cleaner formulation logic—also come with a responsibility: the stronger and more concentrated the extract, the more important it becomes to define a justified dosage range. Standardization improves consistency, but it does not eliminate the need for careful safety review.
5% vs 10% Withanolides: Does Higher Potency Affect Safety?
From a technical standpoint, questions around ashwagandha extract 5% withanolides versus high potency ashwagandha extract forms are really questions about exposure. A higher-potency extract may allow a lower serving weight, but it also changes the active-compound density of the formula. That means withanolide percentage should not be treated as a marketing number alone; it is directly relevant to tolerability, formulation balance, and intended user group. While NIH sources summarize dose ranges from human studies, they do not establish a blanket rule that higher withanolide percentages are universally preferable or equally safe across all applications.
For that reason, discussions around withanolides safety should focus on total daily exposure, extract characterization, and consistency of manufacturing rather than potency in isolation. In responsible supplement development, higher standardization may be appropriate, but only when it is paired with a clearly justified serving size and a clean analytical profile.
Safety Risks Linked to Low-Quality Ashwagandha Extract Powder
Heavy Metals and Contamination Risks
When safety concerns arise around botanicals, the problem is not always the plant itself. In many cases, it is the quality of the finished ingredient. For ashwagandha extract powder, one of the clearest concerns is contamination risk, particularly in poorly controlled supply chains. This is why the discussion around heavy metals herbal extract quality is so important in real-world product development. A material that is not adequately screened for heavy metals, microbiological burden, or contaminants may carry risks that have little to do with the intended extract profile. Federal agencies also remind users that dietary supplements are not approved by FDA before marketing, placing responsibility for safety and labeling on manufacturers and distributors.
In practice, concerns about contaminated ashwagandha are part of a broader issue seen across unsafe herbal supplements generally: when traceability is weak, the final product may not reflect the quality claims made on the label. That is one reason why COA review, contaminant testing, and source verification should be treated as baseline requirements rather than optional documentation.
Inconsistent Withanolide Content
A second major issue is compositional inconsistency. If an extract is poorly standardized, formulators may be working with material that varies from batch to batch in active content, which creates both efficacy and safety problems. This is the core risk behind inconsistent extract quality: the nominal dosage may stay the same while the actual phytochemical exposure changes.
For that reason, non standardized herbal extract materials are harder to position responsibly in supplements that need dependable performance and tolerability. Extract potency variation is not only a formulation challenge; it also weakens the credibility of any safety discussion because it becomes difficult to know what a consumer is really taking from one lot to the next. In our view, this is precisely why standardization should be treated as part of safety architecture, not merely part of commercial positioning.
Residual Solvents and Processing Concerns
Processing quality also matters. For botanicals produced through extraction, the conversation around solvent residue herbal extract control is inseparable from the conversation around purity. A technically sound extract should be manufactured under conditions that manage solvent choice, removal, and verification appropriately. Although the NIH fact sheets focus mainly on clinical safety and interactions rather than production chemistry, they consistently point back to product quality and the limitations of supplement regulation as important background factors in safety assessment.
From a manufacturing standpoint, extraction safety standards and broader herbal extract processing discipline are what separate a credible, application-ready ingredient from a commodity material of uncertain profile. For ashwagandha extract powder, a strong safety position begins with analytical control: identity, standardization, contaminants, residues, and batch reproducibility should all be part of the specification strategy before the ingredient ever reaches a finished formula.
Quality Standards That Ensure Safe Ashwagandha Extract Powder
Ensuring the safety of ashwagandha extract powder begins with clearly defined quality standards at both the raw material and manufacturing levels. In modern supplement development, safety is closely tied to analytical control, regulatory compliance, and full traceability across the supply chain.
COA, Third-Party Testing, and Traceability
A complete ashwagandha extract COA forms the baseline for evaluating extract quality. It typically includes:
•Withanolide standardization
•Microbiological limits
•Heavy metal testing
•Residual solvent analysis
In addition to internal quality control, working with a third party tested herbal extract ensures that specifications are independently verified. This is particularly important for brands developing products for international markets, where regulatory expectations are increasingly aligned with transparency and documentation.
At CHEN LANG BIO TECH, each batch of ashwagandha extract powder is supported by full analytical documentation and traceability, allowing customers to verify consistency from raw material sourcing through to finished extract production.
GMP Manufacturing, HACCP, and International Certifications
Manufacturing standards are a critical factor in ensuring extract safety. Facilities operating under structured systems such as HACCP and ISO-based food safety frameworks provide a more controlled production environment.
For example, our production system is supported by internationally recognized certifications, including:
•HACCP system certification (food safety hazard control)
•ISO 22000 food safety management system
•FDA-registered food facility compliance for export markets
These frameworks ensure that ashwagandha extract powder manufacturer processes—from raw material intake to extraction and final packaging—are managed under standardized safety protocols.
In practical terms, this reduces risks related to contamination, process variability, and quality inconsistency, all of which directly affect product safety.
Global Compliance: Halal, Kosher, and Market Access
Beyond production safety, compliance with global certification standards plays an increasingly important role in ingredient selection.
CHEN LANG BIO TECH supports international formulation needs by providing:
•Halal certification for Islamic markets
•Kosher certification for global dietary compliance
Notably, our Kosher certification covers a wide range of botanical extracts, including ashwagandha extract, confirming its suitability for certified applications
These certifications are not simply formalities. They reflect controlled sourcing, clean processing, and compliance with specific dietary and regulatory requirements, making them highly relevant for brands targeting global distribution.
Regulatory Compliance and Licensed Production
In addition to international certifications, licensed production is a key indicator of manufacturing legitimacy and safety control.
Our facilities operate under officially approved food production licensing, covering plant extracts and related ingredients
This ensures that ashwagandha extract supplier operations comply with national food safety regulations, including:
•Approved production scope
•Controlled manufacturing environment
•Regulatory oversight
For customers, this provides an additional level of assurance that the material is produced within a legally compliant and quality-controlled framework.
Batch Consistency and Supply Reliability
For supplement manufacturers, consistency is as important as safety. Variability in active compounds or physical properties can directly impact formulation performance.
A qualified ashwagandha extract supplier should be able to ensure:
•Stable withanolide levels across batches
•Consistent extract characteristics
•Reliable documentation for every shipment
At CHEN LANG BIO TECH, standardized extraction processes and controlled raw material sourcing allow for consistent batch output, supporting long-term product stability for customers sourcing bulk ashwagandha extract supplier volumes.
Ashwagandha Extract Powder in Supplement Applications: Safe Use Cases
The safety of ashwagandha extract powder is also closely linked to how it is applied in formulations. Different use cases require different dosage strategies, extract specifications, and formulation considerations.
Stress and Sleep Formulations
Ashwagandha is widely used in products targeting stress management and sleep quality. In these formulations, it is typically positioned as an adaptogen that supports relaxation rather than acting as a direct sedative.
Common applications include:
•Stress-support capsules
•Sleep optimization blends
•Relaxation-focused functional products
In these contexts, ashwagandha for stress and sleep supplement ingredients are often combined with other compounds such as magnesium or L-theanine to create balanced formulations.
Sports Nutrition and Recovery Products
In sports nutrition, ashwagandha extract is increasingly used to support recovery, endurance, and overall performance.
Applications include:
•Post-workout recovery formulas
•Adaptogen-based performance supplements
•Testosterone-support blends
As part of ashwagandha sports nutrition strategies, it is often combined with amino acids, creatine, or electrolytes. Its role in recovery supplement ingredients reflects a broader shift toward ingredients that support both physical and physiological recovery.
Daily Wellness and Adaptogen Blends
Ashwagandha extract is also commonly used in general wellness products designed for daily consumption.
These include:
•Adaptogen blends
•Immune and vitality supplements
•Functional beverages
As part of adaptogen supplement ingredients, it is often paired with other botanicals to support overall balance and resilience. In these applications, dosage consistency and extract standardization are especially important for long-term use.
FAQ: Ashwagandha Extract Safety and Usage
Is ashwagandha extract powder safe for daily use?
Short-term use of ashwagandha extract powder is generally considered well tolerated when used within established dosage ranges. However, long-term safety requires careful consideration of dosage, extract standardization, and individual health conditions.
What are the side effects of ashwagandha extract?
Reported ashwagandha extract side effects are typically mild and may include digestive discomfort or drowsiness. Rare cases of more serious adverse effects have been reported, emphasizing the importance of quality and proper usage.
How to ensure the quality of ashwagandha extract powder?
Quality can be verified through ashwagandha extract COA, third-party testing, and supplier transparency. Consistent standardization and contaminant testing are key indicators of a reliable product.
Does higher withanolides content increase risk?
Higher potency extracts do not necessarily increase risk, but they require more precise dosage control. The relationship between withanolides safety and dosage should always be evaluated within the context of the specific formulation.
Where to source safe ashwagandha extract powder in bulk?
Safe sourcing typically involves working with an experienced ashwagandha extract supplier or ashwagandha extract powder manufacturer capable of providing standardized extracts, full documentation, and consistent supply.
Conclusion: Ensuring Safety and Quality in Ashwagandha Extract Applications
The safety of ashwagandha extract powder is determined by a combination of factors, including dosage, standardization, manufacturing quality, and application context.
As the use of adaptogenic ingredients continues to expand, the importance of consistent extract quality and transparent supply chains becomes increasingly evident. Safety is not defined by a single parameter, but by the integration of controlled processes, reliable sourcing, and well-designed formulations.
At CHEN LANG BIO TECH, production of ashwagandha extract powder is managed through standardized extraction systems, controlled raw material sourcing, and batch-level verification to ensure consistency across applications.
For technical documentation or formulation-related inquiries, communication can be directed to:
📧 Email: admin@chenlangbio.com
🌐 Website: www.chenlangbio.com
📚 References
•National Institutes of Health (NIH) – Office of Dietary Supplements
•National Center for Complementary and Integrative Health (NCCIH)
•Clinical studies on Withania somnifera extract and adaptogens
•Regulatory and quality standards for herbal extracts
Send Inquiry
Related Industry Knowledge
- Spirulina vs Phycocyanin: Composition, Applications, and Market Value
- Is PQQ Safe for Daily Use in Supplement Formulations?
- Ajuga turkestanica Extract Powder How to Use It in Sports Formulations
- L-Carnosine: The Safe and Effective Dipeptide for Immune and Gut Health
- Unlocking the Pharmacological Potential of Myricetin Against Various Pathogenesis
- Can D-Luciferin Sodium Salt Be Used for in Vivo Imaging?
- Top 5 Benefits of Witch Hazel Leaf Extract for Skin
- Can You Take EGCG Everyday
- Does Bakuchiol have Any Side Effects
- Is Glutathione GSH Powder Good for Skin Whitening