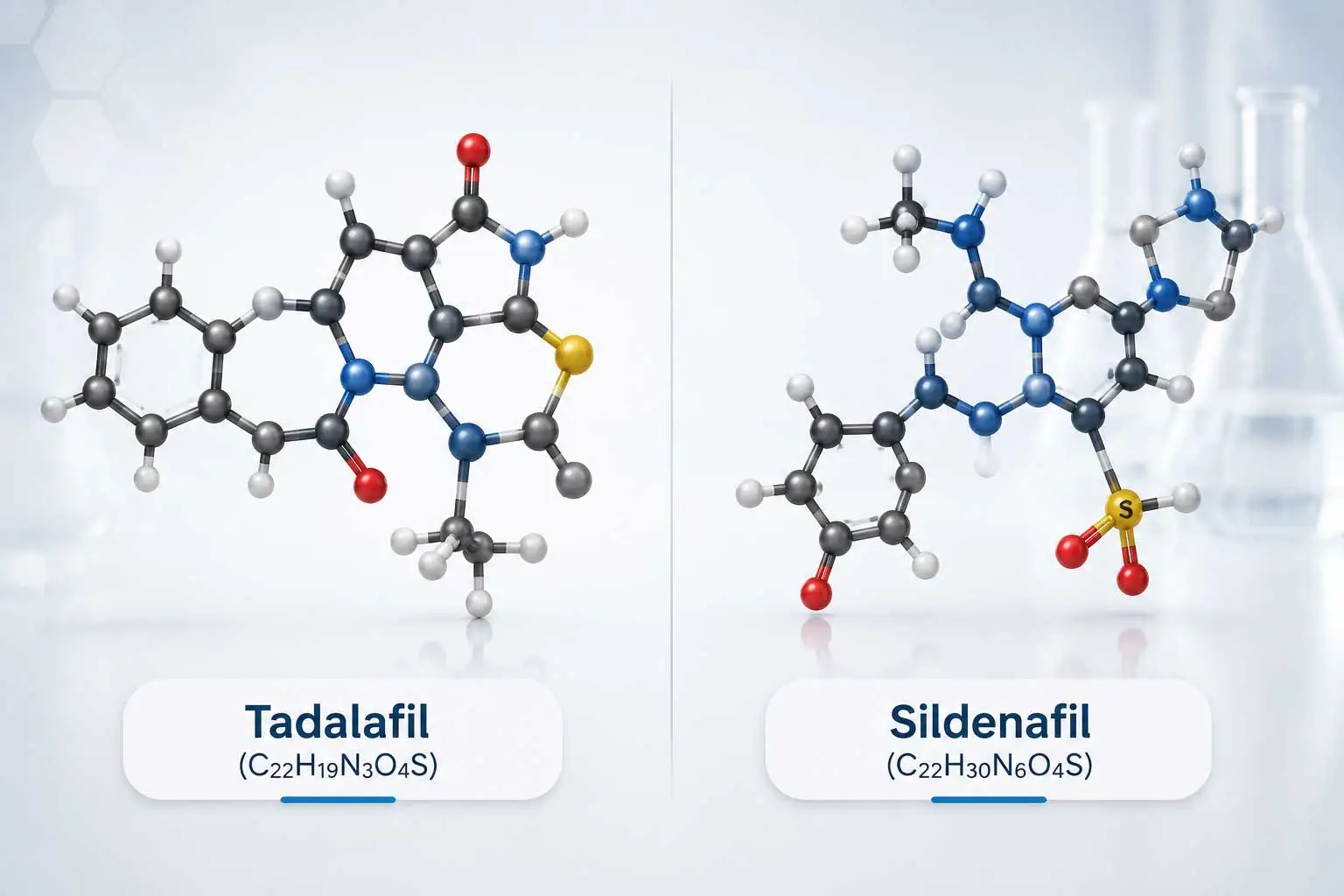
Tadalafil Powder vs Sildenafil Powder: Key Differences and Bulk API Sourcing
2026-05-14 15:46:12
Introduction
The pharmaceutical landscape for PDE5 inhibitors continues to grow, driven by the demand for high-quality APIs used in various formulations. Two of the most widely recognized active pharmaceutical ingredients are tadalafil powder and sildenafil powder. While both play important roles in the development of erectile dysfunction and pulmonary arterial hypertension therapies, their chemical properties, pharmacological profiles, and industrial applications differ significantly.
At CHEN LANG BIO TECH, we provide pharmaceutical-grade tadalafil and sildenafil powders for bulk procurement, emphasizing purity, stability, and regulatory compliance. This article offers an in-depth comparison of tadalafil powder vs sildenafil powder, exploring their key differences, industrial and research applications, and practical guidance for sourcing bulk API from reliable suppliers. Procurement teams, formulators, and R&D professionals can leverage these insights to make informed decisions without compromising on quality or compliance.
Overview of Tadalafil and Sildenafil Powders
Understanding the basic properties of tadalafil powder and sildenafil powder is essential for selecting the right API for pharmaceutical applications. Both compounds are classified as PDE5 inhibitors, yet their molecular structures, pharmacokinetics, and industrial handling requirements are distinct.
Tadalafil Powder Overview
Tadalafil powder is a stable, high-purity active pharmaceutical ingredient (API) used primarily in formulations targeting erectile dysfunction and pulmonary hypertension. The API typically meets pharmaceutical-grade standards, with purity exceeding 99% and stringent controls on particle size, moisture, and stability.
|
Name |
Tadalafil Powder |
|
Other Name |
Calais/ Cialis |
|
Purity |
99.5% |
|
CAS |
171596-29-5 |
|
Molecular Formula |
C22H19N3O4 |
|
Molecular Weight |
389.4 |
|
Appearance |
White fine powder |
|
Package |
25Kg/Paper drum, 1Kg/Aluminum foil bag |
Key points for procurement include:
•Bulk tadalafil powder is supplied under GMP-certified conditions.
•Stability in varying storage conditions makes it suitable for international shipping.
•Documentation including certificate of analysis (CoA) is provided to ensure transparency.
These factors are critical for international procurement and long-term manufacturing reliability.
Sildenafil Powder Overview
Sildenafil powder serves as another widely used PDE5 inhibitor API, with applications in erectile dysfunction treatment and related pharmaceutical research. Like tadalafil powder, it is available in pharmaceutical-grade bulk form, adhering to GMP standards and analytical verification protocols.
Procurement considerations for sildenafil API include:
•Purity and particle size uniformity to ensure formulation consistency.
•Bulk packaging optimized for international shipping and safe handling.
•Availability of technical documentation to support regulatory compliance.
Key Differences Between Tadalafil and Sildenafil
Although tadalafil powder and sildenafil powder share a classification as PDE5 inhibitors, their chemical structure, pharmacokinetics, and industrial applications differ, influencing selection for pharmaceutical formulations. The following table summarizes the primary distinctions:
|
Feature |
Tadalafil Powder |
Sildenafil Powder |
|
Chemical Structure |
[C22H19N3O4] |
[C22H30N6O4S] |
|
Onset of Action |
~30–60 min |
~30–60 min |
|
Duration of Effect |
Up to 36 hours |
~4–6 hours |
|
Solubility |
Slightly soluble in water |
Slightly soluble in water |
|
Storage Conditions |
Stable under controlled humidity, ≤25°C |
Sensitive to moisture and temperature, ≤25°C |
|
Applications |
Erectile dysfunction, pulmonary hypertension |
Erectile dysfunction, pulmonary hypertension |
|
Bulk Procurement Considerations |
Stable for international shipping, GMP compliance, CoA provided |
Requires careful storage during transit, GMP compliance, CoA provided |
Mechanism and Duration Differences
Both tadalafil powder and sildenafil powder function as selective PDE5 inhibitors, preventing the breakdown of cyclic GMP in target tissues. This property underpins their use in pharmaceutical formulations. The primary operational difference lies in duration:
Tadalafil powder provides a longer-lasting effect, up to 36 hours, which may influence product formulation and dosing strategies.
Sildenafil powder has a shorter duration, typically 4–6 hours, necessitating different scheduling in therapeutic applications.
While these differences are primarily of scientific interest, they also guide bulk API procurement, as suppliers must ensure consistent pharmacological properties across batches.
Solubility, Stability, and Storage
The stability and solubility profiles of these APIs are critical for manufacturers and international buyers. Tadalafil powder demonstrates enhanced thermal and humidity stability, making it favorable for long-distance shipping in bulk quantities. In contrast, sildenafil powder requires stricter humidity control and temperature monitoring during storage.
Both APIs are supplied with detailed handling and storage instructions, which is essential for pharmaceutical-grade bulk procurement. Buyers must ensure their warehouses and logistics partners can comply with these standards to maintain high-purity API integrity.
Industrial and Research Applications
Both tadalafil powder and sildenafil powder are widely used in:
•Formulation into oral dosage forms (tablets, capsules)
•Research on PDE5 inhibitors
•Combination therapy studies for pulmonary arterial hypertension
Selection between the two often depends on:
•Desired pharmacokinetic profile
•Stability and shipping conditions
•Procurement volume and batch consistency
For international buyers, sourcing from a trusted bulk API supplier ensures that tadalafil and sildenafil powders meet both quality and regulatory standards.
Quality Standards and Compliance
Ensuring the quality of tadalafil powder and sildenafil powder is critical for manufacturers and research facilities that rely on pharmaceutical-grade bulk API. Both APIs must meet rigorous GMP standards and undergo comprehensive analytical testing to guarantee batch-to-batch consistency.
Purity and Testing
High-purity tadalafil API and sildenafil API are evaluated for chemical identity, purity, moisture content, and particle size. Certificates of Analysis (CoA) provided by trusted bulk API suppliers confirm that each batch adheres to pharmaceutical-grade specifications.
|
Parameter |
Tadalafil Powder |
Sildenafil Powder |
|
Purity |
≥99% |
≥99% |
|
Particle Size |
20–50 μm |
15–45 μm |
|
Moisture Content |
≤0.5% |
≤0.5% |
|
Storage Stability |
24 months @ 25°C |
18–24 months @ 25°C |
Accurate testing and quality assurance protocols are vital for international procurement, ensuring that the bulk powder performs reliably in downstream formulation processes.
Regulatory and International Standards
Both tadalafil powder and sildenafil powder are expected to comply with international pharmaceutical regulations. Suppliers often maintain ISO or GMP certification, supporting cross-border API export. Procurement teams should verify:
•Compliance documentation (CoA, batch records)
•Regulatory approvals for destination countries
•Shipping and storage protocols that align with pharmaceutical-grade API handling
Maintaining adherence to these standards not only safeguards product integrity but also supports long-term reliability in industrial and research applications.
How to Source Bulk Tadalafil and Sildenafil Powders
Sourcing bulk tadalafil powder or sildenafil powder requires careful evaluation of supplier capabilities, regulatory compliance, and logistical considerations. International procurement teams prioritize GMP-certified suppliers who provide technical documentation and consistent supply.
Choosing a Reliable Supplier
When selecting a bulk API supplier, consider:
•Proven experience supplying tadalafil API and sildenafil API internationally
•GMP certification and adherence to quality standards
•Capacity to deliver consistent high-purity powder in required quantities
A trusted supplier ensures smooth production continuity and minimizes supply chain disruptions.
Requesting Samples and Quotes
Most international procurement processes begin with sample requests. Suppliers typically provide small quantities of API for testing and verification before bulk purchase. Clear communication regarding API specifications, purity, and packaging is essential for obtaining accurate quotations.
Shipping, Storage, and Handling Considerations
Both tadalafil powder and sildenafil powder require careful handling, storage, and shipping. Key considerations include:
•Temperature and humidity control during transport
•Appropriate packaging for bulk powder
•Documentation for customs compliance and international regulations
Working with suppliers who provide detailed handling instructions and compliance support simplifies international procurement and reduces the risk of damage or contamination during transit.
FAQ
Q1: What is the difference between tadalafil powder and sildenafil powder?
A1: Both tadalafil powder and sildenafil powder are PDE5 inhibitor APIs, but they differ in duration, stability, and industrial handling. Tadalafil provides a longer effect (up to 36 hours), while sildenafil has a shorter duration (~4–6 hours). Both are supplied in pharmaceutical-grade bulk forms, suitable for research and formulation.
Q2: How do I choose a reliable supplier for bulk tadalafil or sildenafil powders?
A2: Select suppliers with GMP certification, consistent high-purity API, experience in international bulk procurement, and the ability to provide detailed Certificates of Analysis (CoA). Transparent documentation and compliance with shipping and storage standards are essential.
Q3: Can tadalafil and sildenafil powders be shipped internationally in bulk?
A3: Yes, both tadalafil powder and sildenafil powder can be shipped internationally when suppliers follow GMP standards, proper packaging, and compliance with export regulations. Buyers must ensure their logistics partners handle the APIs under recommended temperature and humidity controls.
Q4: What quality documentation should I expect from a trusted API supplier?
A4: A reliable supplier provides CoA, batch records, and analytical testing reports confirming purity, particle size, moisture content, and stability. For international buyers, documentation supporting regulatory compliance is also critical.
Q5: Are there differences in storage conditions between tadalafil and sildenafil powders?
A5: Yes, tadalafil powder generally has higher stability under varying humidity and temperature conditions, while sildenafil powder requires stricter humidity and temperature control. Both APIs must be stored according to pharmaceutical-grade handling standards.
Conclusion
In conclusion, tadalafil powder and sildenafil powder are both essential pharmaceutical-grade APIs with distinct properties that influence their industrial and research applications. Understanding their key differences, stability, and procurement requirements helps procurement teams make informed decisions.
For international buyers seeking bulk tadalafil or sildenafil powders, selecting a reliable, GMP-certified supplier ensures consistent quality, compliance, and smooth logistics. At CHEN LANG BIO TECH, we provide high-purity tadalafil and sildenafil APIs, complete with technical documentation and guidance on handling, storage, and bulk procurement.
For inquiries, sample requests, or quotations, interested parties can reach us via email at admin@chenlangbio.com or visit our website at www.chenlangbio.com for additional information. Our approach supports informed, professional decision-making while maintaining quality and transparency in every transaction.
References
1,Giuliano, F., & Burnett, A. L. (2015). Pharmacology and clinical applications of PDE5 inhibitors. Journal of Sexual Medicine, 12(6), 1495–1507.
2,Pfizer Inc. (2021). Sildenafil Citrate (Viagra) – Product Information. Retrieved from https://www.pfizer.com/products
3,Eli Lilly and Company. (2020). Tadalafil (Cialis) – FDA Prescribing Information. Retrieved from https://www.fda.gov
4,Burnett, A. L., & Nehra, A. (2016). Comparative efficacy and safety of PDE5 inhibitors: tadalafil vs sildenafil. International Journal of Clinical Practice, 70(8), 649–657.
5,CHEN LANG BIO TECH. (2026). Tadalafil and Sildenafil Bulk API Specifications and Procurement Guide. Internal Technical Report, CHEN LANG BIO TECH.
Send Inquiry
Related Industry Knowledge
- How Pyrroloquinoline Quinone Powder Is Used in Dietary Supplements: Applications and Industry Reference Levels
- PQQ in Cognitive Research and Supplement Formulation: An Ingredient Perspective
- How Can Acetylcysteine Powder Improve Your Respiratory Health Formulations?
- What Are the Key Benefits of Choosing High-Purity Indole-3-methanol Powder?
- What are the Benefits of Cetyl Tranexamate HCL in Skincare?
- What are the Benefits of Cetyl Tranexamate HCL?
- Pure Fisetin for Brain Health and Memory Boost
- Premium Haematococcus Pluvialis Extract for Optimal Health Benefits
- Is It Safe to Take Pure Fisetin Powder Every Day
- Quaternium 73 for Acne